Phase I Clinical Trials Unit - Malaga Regional University Hospital
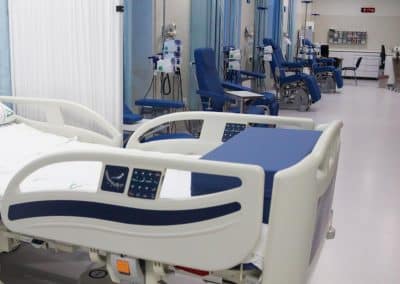
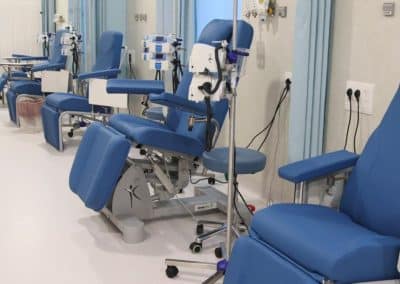
The Phase I Clinical Research Unit of the Regional University Hospital of Málaga is physically located on the first floor of Pavilion 8 at the Hospital Civil. The unit features modern, high-capacity facilities with immediate access to key hospital resources such as the ICU. Its spaces include a patient reception area, nursing control station, three consultation rooms, a day hospital, workrooms, and a technical room, among others.
Responsables de la Unidad

Francisco Carabantes Ocón
Coordinator
He holds a PhD in Medicine and Surgery and has been an oncologist since 1995 at the Regional University Hospital of Málaga (HRUM), where he has also served as Head of the Oncology Department. He has been a tutor for medical residents and actively participates in national and international clinical trials, with a research focus mainly on breast cancer. Additionally, he is a member of scientific societies such as SAOM and SEOM, as well as cooperative research groups on breast cancer (GEICAM, SOLTI) and germ cell tumors. He is also a researcher with the IBIMA group B-22 "Emerging Therapies in Tumor Anti-Adaptation."
francisco.carabantes.sspa@juntadeandalucia.es

Clara María Moreno Bueno
Galería
Factores clave
¿Cuáles son los factores clave de nuestra Unidad?
1. Infraestructuras de un centro de primer nivel
The new Phase I Clinical Trials Unit is part of the healthcare complex of the Regional University Hospital of Málaga, a top-tier center serving a reference population of over 1.5 million people, providing access to a large study population. In addition, the hospital has more than 43 clinical departments.
Strategically located in Pavilion 8 of the Hospital Civil, the unit provides immediate access to key hospital resources, such as the ICU, which is just five meters away. This proximity ensures a rapid, safe, and effective response for patients in early-phase trials.
The Unit has the infrastructure and resources necessary to conduct high-quality clinical research. Featuring modern, high-capacity facilities, it includes areas such as a patient reception area, nursing control station, three consultation rooms, a day hospital, workrooms, and a technical room, among others. All spaces have been designed to meet the technical and functional requirements specific to this type of trial and allow for the exploration of multiple therapeutic areas.
Contract Management Model
IBIMA Plataforma BIONAND has extensive experience in clinical trials, offering comprehensive management that covers all the services required for their development and execution. It provides full support throughout all phases of the study through a management model that ensures agility, flexibility, and a significant reduction in response times. In addition, it has its own functional management units that strengthen its operational capacity.
Early Access to Innovative Therapies
The hospital is accredited for the use of CAR-T cell therapies, establishing itself as a reference center in their application and enabling more patients to access pioneering treatments.
Highly Experienced Team
The purpose of this initiative is to support the development of research lines conducted by emerging and associated groups whose members demonstrate relevant and continuous research activity.
Operational Reliability and Commitment
We ensure strong collaboration with sponsors, based on rapid trial activation, high activity volume, and the achievement of established objectives.
Accreditations and Quality Standards
Indicadores clave

Dossier informativo
Contact
Email:
unidadfase1.hrum@ibima.eu
Phone
951038234 / 618505729
Dirección:
Hospital Regional Universitario de Málaga – Hospital Civil. Pabellón 8, Planta 1ª, Avda. del Arroyo de los Ángeles, s/n, 29009, Málaga.